AIVIS Selected for Government’s "DIPS 1000+" Deep Tech Project to Advance ADC Biomarker Quantification
etnews
April 21, 2026
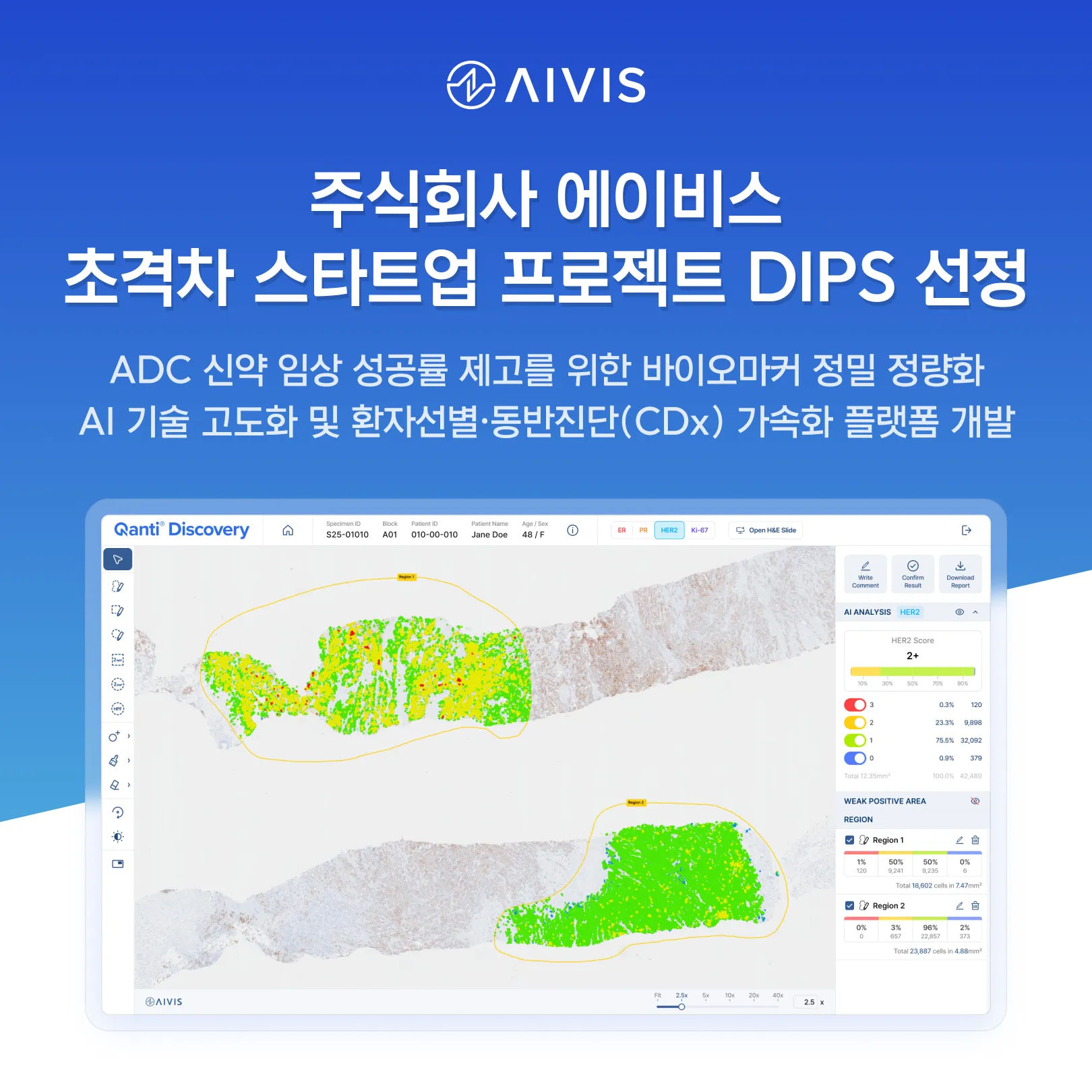
Advancing precision biomarker analysis to enhance ADC clinical success rates | Accelerating development of AI-driven patient selection and Companion Diagnostics (CDx) platform AIVIS (CEO Daehong Lee), a leader in AI-powered pathology image analysis, announced that it has been selected for the "DIPS (Deeptech Incubator Project for Startup) 1000+" initiative in the Life Sciences and Drug Development category. The project is organized by the Ministry of SMEs and Startups. The DIPS 1000+ Project is a national initiative designed to discover and nurture deep-tech startups leading future industries such as Bio-Health and AI. The bio-sector (Life Sciences and Drug Development) is managed by the Korea Institute of Toxicology (KIT). As a selected participant, AIVIS will receive up to 600 million KRW (approx. $450,000) over three years to support technology commercialization and R&D. With this selection, AIVIS plans to accelerate the expansion of its clinical-focused AI technology across the entire drug development and clinical trial lifecycle. The company will focus on strengthening its AI-based quantitative analysis capabilities for biomarker evaluation, patient selection, and companion diagnostics (CDx)—all of which are critical components in the development of Antibody-Drug Conjugates (ADCs). AIVIS provides high-precision AI solutions that quantitatively analyze biomarker expression in pathology images. Its flagship product, "Qanti IHC," is an AI-based software medical device that performs cell-level quantitative analysis of immunohistochemistry (IHC)-stained slides; it received approval from the Ministry of Food and Drug Safety (MFDS) in September 2024. Furthermore, its "Qanti Discovery" R&D platform quantitatively analyzes pathology data generated during drug development and clinical trials, supporting biomarker-based patient selection and data-driven decision-making. AIVIS has achieved significant milestones in the AI pathology field through rigorous real-world clinical validation. Currently, Qanti IHC is utilized by pathologists in over 10 major hospitals across Korea. The company offers AI solutions optimized for clinical workflows through high compatibility with global digital pathology scanner leaders, including Philips, Leica, and Roche. Building on its clinical success, AIVIS is expanding into the pharmaceutical R&D domain. In February 2026, the company signed a joint research agreement with AimedBio, a specialist in ADC drug development, and secured a strategic investment. The two companies are currently utilizing Qanti Discovery to analyze pathology data generated during ADC development, aiming to advance precision biomarker-based patient selection strategies. CEO Daehong Lee stated: "Building upon our AI technology validated in clinical settings, AIVIS is evolving into a biomarker quantitative analysis platform applicable across the entire drug development lifecycle. We will continue to deepen our partnerships with global pharmaceutical and biotech companies to contribute to the advancement of data-driven precision medicine and global drug development innovation."